 Discovery Information Discovery Information
|
| Who: Sir Humphrey Davy |
| When: 1807 |
| Where: England |
|
 Name Origin Name Origin
|
| From potash (pot ash); K from latin: kalium |
| "Potassium" in different languages. |
|
 Sources Sources
|
| Found in minerals like carnallite [(KMgCl3).6H2O] and sylvite (KCl). Potassium makes up about 2.40% of the weight of the Earth's crust, it is the seventh most abundant element.
The main sources of potash are mined in USA (California, New Mexico and Utah) and Germany. Around 200 tons are produced annually.
|
|
 Abundance Abundance
|
| Universe: 3 ppm (by weight) |
| Sun: 4 ppm (by weight) |
| Carbonaceous meteorite: 710 ppm |
| Earth's Crust: 15000 ppm |
| Human: |
| 2 x 106 ppb by weight
|
| 3.2 x 105 ppb by atoms
|
|
 Uses Uses
|
| Used as potash (potassium carbonate) in making glass, soap, lenses and salt substitute. Also as potassium nitrate (KNO3, also called saltpeter) it is used to make explosives and to colour fireworks in mauve.
|
| Glass treated with liquid potassium is much stronger than regular glass. |
| Potassium chloride (KCl) is used as a substitute for table salt and is also used to stop the heart, e.g. in cardiac surgery
and in executions by lethal injection in solution.
|
|
 History History
|
| Potassium was discovered in 1807 by Sir Humphry Davy, who derived it from caustic potash (KOH). Potassium was the first metal that was isolated by electrolysis.
|
|
 Notes Notes
|
| Potassium is the second least dense metal; only lithium is less dense. It is a soft, low-melting solid that can easily be
cut with a knife. Freshly cut potassium is silvery in appearance, but in air it begins to tarnish toward grey immediately.
|
| Potassium is a necessary mineral in daily nutrition; it assists in muscle contraction and in maintaining fluid and electrolyte
balance in body cells.
|
| Formerly called kalium hence the symbol 'K'. |
|
 Hazards Hazards
|
| Solid potassium reacts violently with water, and should therefore be kept under a mineral oil such as kerosene and handled
with care. When in water, it may catch fire spontaneously, burning with a purple flame. Unlike lithium and sodium however, potassium cannot be stored under oil indefinitely. If stored longer than 6 months to a year, dangerous shock-sensitive
peroxides can form on the metal and under the lid of the container that can detonate upon opening.
|
| Potassium is very harmful by ingestion or through the skin or eye contact, it may cause irreversible eye damage. |




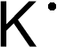
 Discovery Information
Discovery Information
 Name Origin
Name Origin
 Sources
Sources
 Abundance
Abundance
 Uses
Uses
 History
History
 Notes
Notes
 Hazards
Hazards
 Images
Images


